Proteins are generally thought of as being soluble with a single well-defined three-dimensional structure. However, some proteins form large aggregates, either for a specific biological function, or as part of a pathological process. Amyloid fibrils are a specific kind of ordered protein aggregate that we are mainly focused on. Our research investigates structural and mechanistic details of these amyloid proteins, as well as revealing what factors influence their conversion to amyloid.
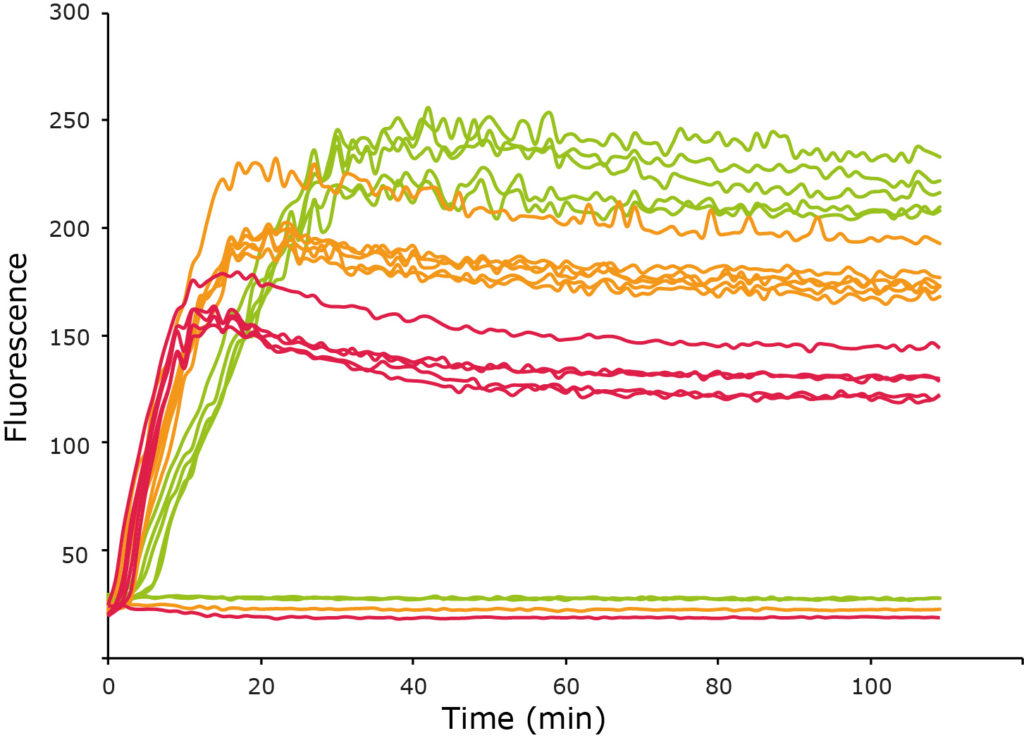
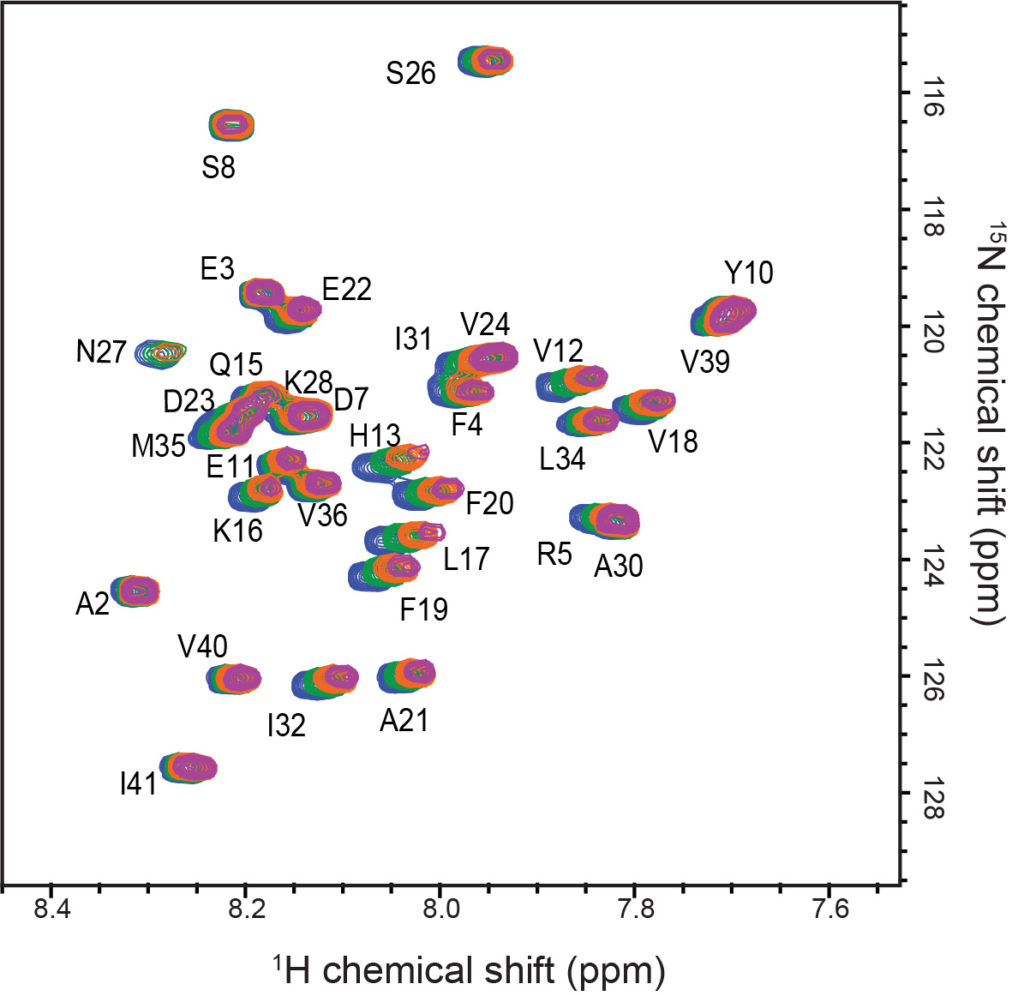
To unravel these questions we combine a range of structural, biochemical and biophysical techniques including NMR spectroscopy (solution and solid state), fluorescence-based aggregation assays and electron microscopy.
Currently our ongoing research is focused on two specific amyloid-forming proteins:
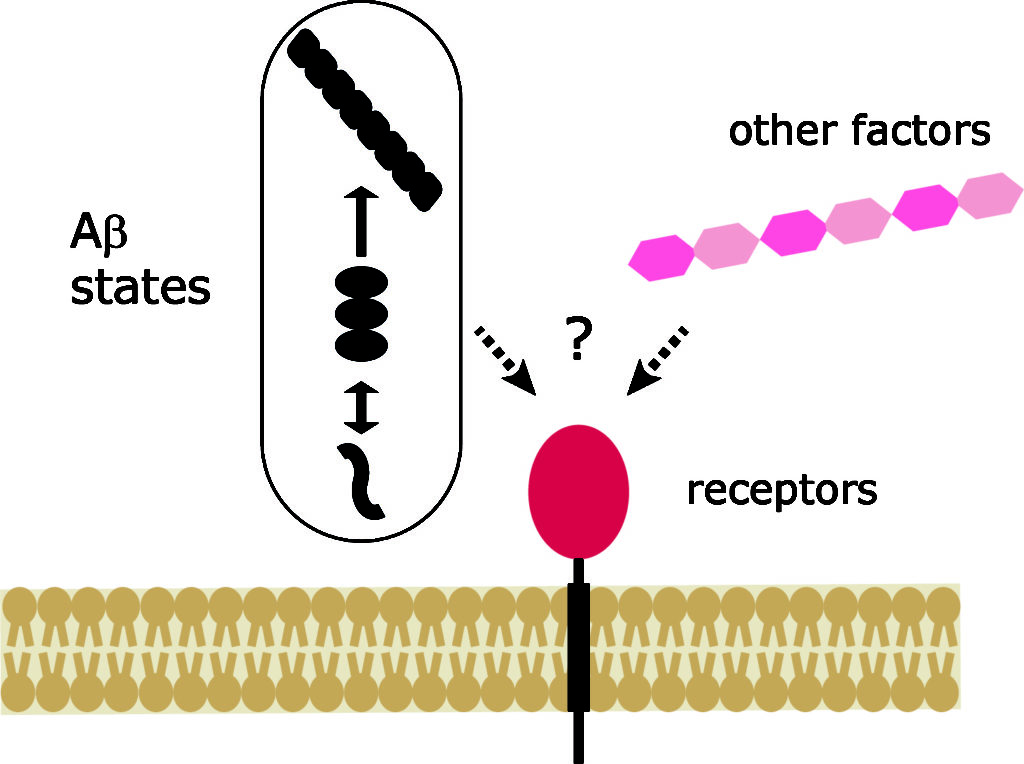
1. The peptide amyloid-beta is involved in Alzheimer’s disease development.
We aim to understand how this protein and it’s different conformational forms are involved in disease development by dissecting its interactions with different brain factors, such as receptors, chaperones and carbohydrates.
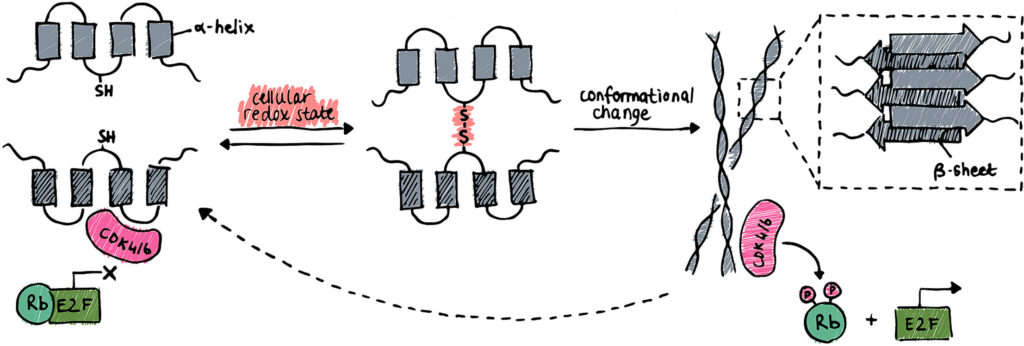
2. The tumour suppressor p16 forms amyloids.
We have recently discovered that p16 (also known as p16INK4A or CDKN2A) can convert to amyloid fibrils under mild oxidizing conditions. This is a fascinating new property of this important cell-cycle controller, that has not been recognised until now. The relevance of this state in cancer and for the function of the protein is still a mystery.